the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
High-frequency NO3− isotope (δ15N, δ18O) patterns in groundwater recharge reveal that short-term changes in land use and precipitation influence nitrate contamination trends
Martin Suchy
Leonard I. Wassenaar
Gwyn Graham
Bernie Zebarth
Poultry manure is the primary cause of nitrate () exceedances in the transboundary Abbotsford–Sumas aquifer (ASA; Canada–USA) based on synoptic surveys two decades apart, but questions remained about seasonal and spatial aspects of agricultural nitrate fluxes to the aquifer to help better focus remediation efforts. We conducted over 700 monthly δ15N and δ18O of nitrate assays, focusing on shallow groundwater (< 5 years old) over a 5-year period to gain new insight on spatio-temporal sources and controls of groundwater nitrate contamination. concentrations in these wells ranged from 1.3 to 99 mg N L−1 (n=1041) with a mean of 16.2±0.4 mg N L−1. The high-frequency 15N and 18O isotope data allowed us to identify three distinctive source patterns: (i) primarily from synthetic fertilizer, (ii) dynamic changes in nitrate due to changes in land use, and (iii) from a mix of poultry manure and fertilizer. A key finding was that the source(s) of nitrate in recharge could be quickly influenced by short-term near-field management practices and stochastic precipitation events, which ultimately impact long-term nitrate contamination trends. Overall, the isotope data affirmed a subtle decadal-scale shift in agricultural practices from manure increasingly towards fertilizer nitrate sources; nevertheless, poultry-derived N remains a predominant source of nitrate contamination. Because the aquifer does not generally support denitrification, remediation of the Abbotsford–Sumas aquifer is possible only if agricultural N sources are seriously curtailed, a difficult proposition due to longstanding high-value intensive poultry and raspberry and blueberry operations over the aquifer.
- Article
(7787 KB) - Full-text XML
-
Supplement
(142 KB) - BibTeX
- EndNote
The global widespread use and over-application of synthetic and manure N nutrients in agriculture have caused widespread groundwater nitrate () contamination in surficial aquifers around the world (Haslauer et al., 2005; Hamilton and Helsel, 1995; Spalding and Exner, 1993). Furthermore, with global trends towards increased agricultural intensification, threats to groundwater quality are correspondingly heightened (Vorosmarty et al., 2000; Böhlke, 2002). In agricultural settings, elevated groundwater concentrations typically reflect a supply of N from manure, synthetic fertilizer, or soil organic matter which exceeds, or is poorly synchronized with, crop N requirements (Canter, 1997). The risk of contamination is especially high in phreatic aquifers comprised of coarse-grained permeable soils with minimal propensity for natural attenuation and remediation processes such as microbial denitrification. Studies have used stable isotopes of nitrate (δ15N, δ18O) to differentiate different source inputs (Mitchell et al., 2003; Wassenaar et al., 2006; Xue et al., 2009; Pastén-Zapata et al., 2014), while others have used δ15N and δ18O to examine the anaerobic remediation of nitrate by microbial denitrification (Böhlke and Denver, 1995; Kellman and Hillaire-Marcel, 2003). Others used isotopes of nitrate to assess soil N transformations (Savard et al., 2010) or temporal variations in agricultural leachate to groundwater (Ostrom et al., 1998; Loo et al., 2017; Savard et al., 2007).
Concentrations of non-agricultural in aquifers that are low (< 1 mg N L−1) and below drinking water standards are usually attributed to atmospheric N deposition, organic N from plant decomposition or land breakage, and geological sources that are mobilized due to disruptions in water recharge fluxes such as introduction of irrigation (Canter, 1997). Choi et al. (2003) suggests low groundwater concentrations consistently below 3 mg N L−1 with δ15N values between +5 ‰ and +8 ‰ are likely derived from natural soil or organic N sources (average δ15N +5 ‰). Loo et al. (2017) reported non-agricultural soil δ15N nitrate ranges between +3.7 ‰ and +4.9 ‰ in our study area (Table 1).
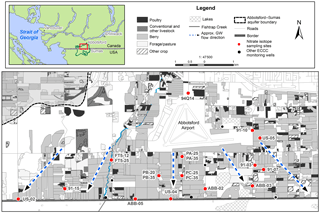
Figure 1Location of the Abbotsford–Sumas aquifer (ASA), southwestern BC, Canada, and northwestern Washington state, USA, along with simplified agricultural land use and sampling locations with groundwater mean residence times (MRT) of < 5 years. Arrows show the approximate groundwater flow direction.
Table 1Local synthetic fertilizer, poultry manure, soil N, and leachate δ15N values used in the Abbotsford area.
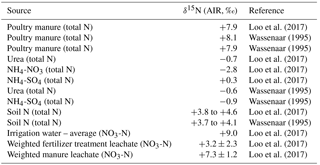
Sources of nitrate from animal waste arise from dispersed agricultural field applications and/or point-source manure storage facilities (liquid and solid). Under aerobic soil conditions, quickly forms from oxidation of after manure application (Aravena et al, 1993). Due to preferential volatilization of 14N in gaseous NH3 from during wet storage and/or application of manure, manure-derived is accordingly enriched in 15N (Kendall, 1998). Nitrate derived from manure or septic waste can have δ15N values between +10 ‰ and +25 ‰, (Wassenaar, 1995; Kreitler, 1975; Heaton, 1986; Aravena and Robertson, 1998), generally revealing little distinctive 15N isotopic resolution between these two organic N waste sources. Poultry manure solids have average δ15N values of approximately +7.9 ‰ in the study area (Loo et al., 2017; Wassenaar, 1995). In North America, urea (CO(NH2)2) (nitrogen, phosphorus, potassium (N-P-K): 46-0-0) is one of the most common forms of synthetic fertilizers used (Overdahl et al., 2007). Other forms of synthetic fertilizers include ammonium nitrate (NH4-NO3) (34-0-0) and ammonium sulfate (NH4-SO4) (22-0-0), as shown in Table 1. Each of these is manufactured by fixation of atmospheric N (δ15N = ∼ 0 ‰), resulting in δ15N values from −2.8 ‰ to +0.3 ‰ (Loo et al., 2017). In the Abbotsford–Sumas aquifer (ASA) area, berry-specific fertilizer blends are commonplace (Table 1), where N is derived from one of the above sources (Loo et al., 2017; Wassenaar, 1995).
The δ18O values of synthetic fertilizer-derived typically range between +18 ‰ and +22 ‰, because the oxygen in nitrate originates from air (δ18O ‰) and 18O-depleted H2O (Amberger and Schmidt, 1987). Nitrate derived from NH4-NO3 fertilizers, where 50 % of the oxygen is from nitrification of NH4 fertilizer and 50 % is from synthetic fertilizer, has reported δ18O values around +13 ‰ (Aravena et al., 1993). In this study, the δ18O of groundwater is relatively uniform ( ‰; Wassenaar, 1995), which would result in δ18O values around zero ‰ for derived only from the nitrification of soil or organic N or ammonium sources.
In the phreatic transboundary Abbotsford–Sumas aquifer (Canada–USA, Fig. 1), long-term nitrate contamination trends and isotopic studies have been conducted over several decades. The isotopic apportionment of sources in the aquifer was based on two summertime synoptic isotopic studies spanning a full decade, which revealed that poultry manure was the predominant source of , with localized long-term shifts towards inorganic fertilizer sources (Wassenaar, 1995; Wassenaar et al., 2006) due to changes in agricultural practices (Zebarth et al., 2015). These agricultural changes include decadal shifts from beef and dairy feed production to lower N uptake berry production, changes in irrigation methods from sprinklers to low-pressure drip fertigation, planting of cover crop for berry alley management, and a slow transition from raspberries to blueberries. One critique of the previous synoptic isotope studies was that sampling (and hence interpretations) was prejudiced to summer snapshots and thereby could be biased, especially for the numerous shallow and highly responsive water table wells spanning the aquifer and winter-biased recharge (Environment Canada, 2014). The seasonal dynamics of sources and fluxes and the potential for isotopic changes due to soil and unsaturated zone cycling were not fully evaluated and need to be considered to improve surface nutrient applications and agricultural management practices.
To address this knowledge gap, we conducted high-frequency (monthly) concentration and isotope analysis of the ASA over a 5-year period, with a focus on shallow wells having groundwater residence times of < 5 years as determined by 3H-He age dating. Our aim was to determine whether high-frequency (monthly) nitrate and isotope (δ15N, δ18O) sampling improved upon previous interpretations of N sources and processes and whether important seasonal changes in the proportion of sources recharging to groundwater were overlooked by synoptic snapshots. Our goal was to gain improved insight on the spatio-temporal sources and controls of groundwater–nitrate dynamics and thereby to help better inform agricultural nutrient management practices and potential remediation efforts in the aquifer.
2.1 Study area and hydrogeologic setting
The Abbotsford–Sumas aquifer is a shallow phreatic transboundary aquifer located in southwestern British Columbia, Canada, and northwestern Washington state, USA (Fig. 1). The ASA is the most intensively studied nitrate-contaminated aquifer in Canada (Zebarth et al., 1998, 2015) and covers an area of about 200 km2, with approximately 40 % of the total surface area in Canada (Cox and Kahle, 1999). Our study area encompassed approximately 40 km2 on the Canadian side of the aquifer, between the Abbotsford International Airport and the Canada–USA border (Fig. 1). Land use on the aquifer is predominantly commercial raspberry and blueberry production, mixed with intensive commercial poultry barn operations (Fig. 1) and is < 5 % rural residential; (BC Ministry of Agriculture, unpublished data).
The aquifer is typically 10–25 m thick but reaches 70 m thickness towards the southeast portion (Cox and Kahle, 1999). The aquifer comprises coarse glacio-fluvial sand and gravel with minor till and clayey silt lenses (Armstrong et al., 1965), with glacio-marine clays confining the aquifer below (Halstead, 1965). The high sand and gravel content results in a high transmittance of water, with mean hydraulic conductivities (K) of m s−1 (Chesnaux and Allen, 2007) to m s−1 (Cox and Kahle, 1999). The thin organic-poor soils (0–70 cm) are medium-textured aeolian deposits, moderately well to well drained, and are classified as Orthic Humo-Ferric Podzols (Luttmerding, 1980).
Average annual precipitation across the aquifer (1981–2010) is 1538 mm, of which 70 % falls between October and March (Environment Canada, 2014). Annual recharge estimates range from 850 to 1100 mm (Zebarth et al., 2015), and water table depths typically vary widely between 2 and 20 m below surface depending on the location and season. Annual water table fluctuations average ∼ 3.6 m (Scibek and Allen, 2006). The overall flow direction in the aquifer is south (Fig. 1), southeast, and southwest at linear velocities of up to 450 m yr−1 (Liebscher et al., 1992; Cox and Kahle, 1999).
The aquifer is highly vulnerable to surface-derived and other contamination because of (i) intensive agricultural activity; (ii) highly permeable soil, coarse sand, and gravel lithology; and (iii) high precipitation amounts in the fall and winter when nutrient uptake by crops is lowest and the leaching potential is greatest (Kohut et al., 1989; Liebscher et al., 1992). Elevated groundwater–nitrate concentrations exceeding drinking water guidelines are observed since the 1970s (Zebarth et al., 2015). Mitchell et al. (2003) and others (Wassenaar et al., 1995) showed vertical stratification of nitrate in groundwater was linked to agricultural practices, with the greatest nitrate concentrations (> 20 mg N L−1) occurring in shallow water table regions (< 10 m b.g.l., metres below ground level), while average groundwater–nitrate concentrations (< 10 mg N L−1) in deep wells (> 10 m screen depth below average static water level) were lower and relatively stable over time. Based on Environment and Climate Change Canada (ECCC) monitoring, the highest seasonal and temporal variations in are found in wells screened near the water table. Both seasonal and long-term temporal variations in groundwater–nitrate over decadal timeframes are well documented (Liebscher et al., 1992; Graham et al., 2015). The aquifer has little widespread intrinsic capacity to sustain microbial denitrification (self-remediation) because of largely aerobic conditions and the low organic content of the aquifer materials (Wassenaar, 1995), but it can occur in localized pockets around riparian discharge zones.
2.2 Sample collection and analysis
Monthly groundwater samples (n=56 per well) were collected from 19 selected monitoring wells from September 2008 to March 2013. These wells were selected based on the following criteria: (1) groundwater having a < 5-year residence time based on 3H-He age dating (Wassenaar et al., 2006), (2) representative spatial coverage within the monitoring network, and (3) wells representing aerobic groundwater where denitrification does not occur (Tesoriero, 2000; Wassenaar et al., 2006). These criteria helped to ensure that high-frequency nitrate and isotopic patterns stem from short-term nitrate responses unaffected by historical or subsurface biogeochemical processes or mixing with deeper water and could therefore be more explicitly linked to contemporary landscape and agricultural activities and practices happening roughly within a 5-year timeframe.
Static water level measurements were taken prior to pumping and were reported in metres above mean sea level (m a.s.l.). Groundwater was sampled from the wells using a Grundfos® stainless-steel submersible pump, Teflon® lined LDPE (low-density polyethylene) tubing, and stainless-steel fittings and valves. Well water was pumped through a flow-through cell housing a calibrated YSI® multi-probe sonde (temperature; pH; specific conductance; oxidation reduction potential, ORP; and dissolved oxygen, DO). General chemistry and isotope water samples were collected after at least three well volumes were purged and the YSI® field parameters were stabilized. All bottles were rinsed three times with sample water prior to filling. Water samples for major ion and nutrient concentrations were taken in 1 L LDPE bottles, filtered through 0.45 µm cellulose acetate membrane filters, stored at 5 ∘C, and analyzed within 5 days for nitrate using standard ion chromatography techniques. Nitrate concentrations were determined at the Pacific and Yukon Laboratory for Environmental Testing in North Vancouver, BC, Canada. Nitrate results are reported as mg N L−1 with a minimum detectable limit of 0.02 mg L−1.
Samples for nitrate isotope analyses (δ15N, δ18O) were field filtered through 0.45 µm cellulose acetate membrane filters and frozen (−40 ∘C) in 125 mL HDPE (high-density polyethylene) bottles. Nitrate isotope assays were conducted by the University of Calgary Stable Isotope Laboratory, using a microbial reduction to N2O described elsewhere (Casciotti et al., 2002; Sigman et al., 2001). All δ15N values are reported relative to the atmospheric air reference (Mariotti, 1983) and normalized by analyzing reference materials IAEA-N3 (δ 15N ‰), USGS32 (δ15N ‰), USGS34 (δ15N ‰), and USGS35 (δ15N ‰) along with samples. The analytical uncertainty for δ15N was ±0.5 ‰. The δ18O values were reported relative to the VSMOW reference (Coplen, 1994) and determined by analyzing reference materials IAEA-N3 (δ18O ‰), USGS32 (δ18O ‰), USGS34 (δ18O ‰), and USGS35 (δ18O ‰). The analytical uncertainty for δ18O was ±1.0 ‰.
Nitrate and chloride concentrations were log-transformed prior to analysis to ensure normal distributions and were evaluated using principal component analysis (PCA) and factor analysis. Statistical analyses (at the 95 % confidence level), including multivariate time series analyses, were conducted using the Kruskal–Wallis methods for determining seasonality, log-normal transformations, Mann–Kendall trend analyses, and Gaussian mixture and Bayesian clustering models using WQHydro®, ProUCL 5®, and XLSTAT® (Lettenmaier, 1988; Thas et al., 1998). Seasonal Mann–Kendall trend analyses were deemed inappropriate for evaluating nitrate concentration seasonality as the repeating periods were correlated to precipitation patterns instead of calendar month and because peak nitrate concentration timings varied from year to year, resulting in a determination of non-seasonality.
3.1 Groundwater nitrate concentrations
Results of monthly nitrate concentrations in the water table wells in the aquifer over the 5-year sampling period ranged from 1.3 to 99.0 mg N L−1 (n=1041), having a mean concentration (± SE) of 16.2±0.1 mg N L−1. Approximately 76 % of the shallow groundwater locations (16 of 19 sites) exceeded the maximum allowable concentration (MAC) of 10 mg N L−1 in the Canadian Drinking Water Guidelines (Health Canada, 2013). These nitrate exceedances were consistent with previous observations of high nitrate concentrations in shallow wells in the aquifer (Hii et al., 1999). Previous studies reported concentrations exceeding the MAC in 58 %, 69 %, and 59 % of wells (Wassenaar, 1995; Zebarth et al., 1998; Wassenaar et al., 2006), respectively. The current study only had ∼ 50 % monitoring wells in common with previous investigations because early studies also sampled deeper monitoring wells containing older groundwater.
A time-series analysis showed that overall concentrations steadily increased from 14.1 to 18.4 mg L−1 in the targeted shallow wells over our 5-year study period, which contrasted with long-term declines observed for a wider depth variety of wells in the Canadian portion of the aquifer (Zebarth et al., 2015). Graham et al. (2015) identified several key drivers causing the short-term (intra- and inter-year) nitrate trends (increases or declines) that contrasted with the long-term (inter-decadal) declines. These key drivers were primarily stochastic rainfall patterns (wet vs. dry years, with first major rainfalls occurring in October) and short-term land use change factors. The overall increasing nitrate trend in the 19 wells could be attributed to the marked increases in concentrations in three of the wells occurring in the second half of our study, without which the average concentration in the remaining 16 wells (15.5 mg L−1) did not change. These nitrate increases were attributed to (i) clearing of an adjacent woodlot, and the subsequent application of large quantities of poultry manure as a soil amendment up-gradient of PC-25 and PC-35 in 2011; and (ii) a raspberry field up-gradient of US-02 that underwent a renovation cycle (described in Zebarth et al., 2015), which likely also included soil organic amendments. Wells 94Q-14, PA-25, and PA-35 did not exceed the nitrate MAC because these sites were located up-gradient of the most intensive agricultural production areas.
Almost half the 19 shallow monitoring wells (47 %) showed seasonality, with maximum concentrations usually occurring in the springtime. Nitrate accumulates in the soil and root zones over the summer, and a large proportion of nitrate flushing to the water table happens with the first major recharge events in the fall and winter rainy season (Kowalenko, 2000). Subsequent recharge typically has lower nitrate concentrations as the availability of dissolved soil nitrate drops. Previous evidence of flushing in the fall is shown by Wassenaar (1995) and Zebarth et al. (1998), when precipitation, recharge rates, and soil- are at their peak. Coupled with vadose zone infiltration lag times of several months, which do not vary significantly between sites (Herod et al., 2015), accordingly peak concentrations reaching the water table are usually observed in the springtime.
All wells have aerobic groundwater (mean = 8.9 mg O2 L−1; Table S1 in the Supplement); however, two sites (ABB-03 and US-02) showed short intervals of lowered DO levels (< 1 mg L−1) in the winter months, coinciding with higher water tables. Chloride levels were on average 8.7±3.0 mg L−1. At six sites (91-10, 91-15, PA-25, PA-35, US-02, and US-05), and Cl concentrations exhibited a covariance (Pearson's R correlation coefficients > 0.5), suggesting similar sources. Three sites (PC-25, FT5-12, and FT5-25) exhibited an offset between and Cl; however, the Cl peaks usually lagged behind peaks by 1–3 months, which is inconsistent with the conservative properties of Cl−. This was also observed by Malekani (2012) and may be caused by differences in timing of and Cl availability in combination with growing season leaching under drip irrigation or variable transit times though the unsaturated zone. The remaining sites exhibited limited seasonal nitrate and chloride variability or showed no correlation between these variables, suggesting more mixing within local groundwater and possibly longer transit times in the unsaturated zone.
3.2 Nitrate N and O isotopes
Overall, the mean (±SE) groundwater nitrate δ15N value from the 19 study wells was ‰ (n=717), which was consistent with positive δ15N values of local poultry manure solid sources (Wassenaar, 1995; Loo et al., 2017) as summarized in Table 1. Higher δ15N source values were observed in nitrate extracted from soil below former manure stockpiles and ranging up to +14 ‰, due to ammonia volatilization (Wassenaar, 1995). Mean nitrate δ18O was ‰ (n=717), which was typical of values derived during the nitrification of manure or synthetic fertilizers (Xue et al., 2009). Previously measured groundwater δ18O in the aquifer ranged between −10 ‰ and −12 ‰ and, coupled with O derived from air (+23.5 ‰), the current nitrate δ18O values were comparable with earlier δ18O values (Wassenaar, 1995) of ‰ (n=16) and ‰ (n=40) a decade later (Wassenaar et al., 2006).
To further assess sources and seasonality of groundwater nitrate from these 19 shallow wells, the results were evaluated using nitrate concentrations and isotopic compositions. A Keeling plot of 1∕ vs. δ15N (Fig. 2a), supported by a Gaussian mixing model, suggests three main nitrate groupings with the following proportions and interpretation: (i) a historical mixing (47 %) trend between high and high δ15N (manure-derived) and intermediate and low δ15N values (fertilizer-derived); (ii) fertilizer and soil N dominant (47 %) low-intermediate NO3 and low δ15N (+2 ‰ to +4 ‰); (iii) intermediate and intermediate δ15N (+8 ‰), with a mixed source of manure or soil and N fertilizer (6 %). A Bayesian VVV clustering model (which considers the volume, shape, and orientation of data points) using δ 15N and suggested five possible groupings (Fig. 2b), with means shown in Table 2. These findings altogether suggest that field-scale agricultural management practices up-gradient of the monitoring wells resulted in four quantifiably distinctive nitrate isotopic clusters (Table 3 – source grouping). The first two Bayesian groups were amalgamated as they suggested the same isotopic source, but with partial isotopic enrichment.
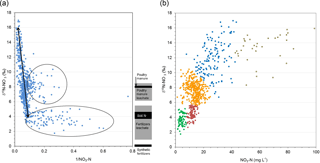
Figure 2(a) Keeling plot of 1∕NO3 (x axis) vs. δ15N (y axis). Three distinct groups are described as follows: (i) arrow represents the mixing line between fertilizer and enriched manure endmembers, (ii) (bottom) wide range NO3 (mineral fertilizer leachate and soil N), and (iii) (middle group) manure–fertilizer mixture. δ15N reference sources (Wassenaar, 1995; Loo et al., 2017). (b) δ15N vs. nitrate Bayesian clustering model suggests five distinct groupings.
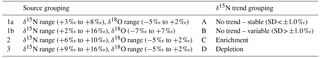
Another clustering approach, based on δ15N trends and seasonality in the 19 wells over the course of the study was also evaluated. In this case, sites were separated into four clusters (Table 3 – trend grouping) as follows: (a) no trend with stable δ15N values (SD < ±1.0 ‰), (b) no trend with variable δ15N values (SD > ±1.0 ‰), (c) 15N enrichment trends, and (d) 15N depletion trends.
3.2.1 Nitrate isotopic variations
Considering the Bayesian and Gaussian clustering approaches altogether, we separated the nitrate and isotope data into four distinctive groups (Fig. 3) based on their isotopic values (three primary groups and one subgroup), both in relation to each other and to well-known sources.
Group 1a groundwater was impacted by synthetic fertilizer and/or organic N and showed little isotopic variability, while group 1b was similar but impacted by clear short-term spikes in δ15N and . Group 2 was dominated by poultry manure with some influence of 15N-depleted sources, while group 3 was dominated solely by poultry manure N.
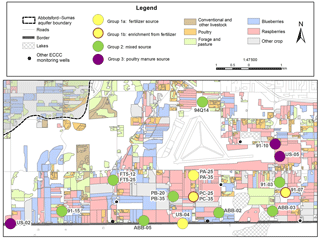
The four wells categorized into group 1a, with δ15N values of +3 ‰ to +8 ‰ representing 21 % of the 19 sites (PA-25, PA-35, 91-07, and US-04), had a mean δ15N value of +5.0 ‰. The N isotope distribution of these samples suggests they were dominated by synthetic fertilizers and natural (background) soil N sources (δ15N of −1.0 ‰ and +4.0 ‰, respectively). Loo et al. (2017) reported that weighted δ15N of fertilizer treatment leachate in the ASA is ‰. Sampling wells in this group did not exhibit large seasonal swings in concentration or δ15N values, although strong seasonality was found for in wells PA-25 and PA-35. These isotope data suggest a combination of annual synthetic fertilizer applications with occasional poultry manure application as a soil amendment, which is a common agricultural practice in this area, particularly with blueberry crops.
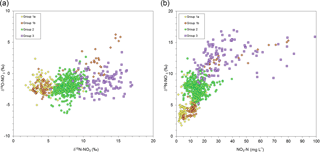
Figure 3(a) Nitrate δ18O vs. δ15N cross plot. (b) Distribution of 19 well sites grouped by δ15N range and δ18O. Group 1a: δ15N range (3 ‰ to 8 ‰). Group 1b: δ15N range (+2 ‰ to +16‰). Group 2: δ15N range (+6 ‰ to +10 ‰). Group 3: δ15N range (+9 ‰ to +16 ‰).
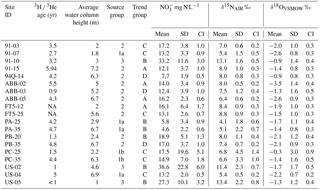
Table 4Results summary with 3H ∕ 3He groundwater ages in years (Wassenaar et al., 2006); average water column height (metres; mid-screen depth below average static water level); isotopic distribution and trend groupings; NO3-N, δ15N, and δ18O values (mean, standard deviation, and confidence intervals (α=0.05)). NA means not available.
The group 1b wells were distinctive because the mean nitrate δ 15N value was more depleted than poultry manure (+6.7 ‰), but spanned a wider δ15N range from +2 ‰ to +16 ‰ and represented only two wells (PC-25 and PC-35). In addition, both exhibited nitrate 18O enrichment, coupled with increasing δ15N values (Fig. 3a) and concentrations. Well PC-25 was likely subjected to localized and temporal root zone denitrification since some δ 18O values increased above +5 ‰; however, groundwater DO values were never below 8.8 mg L−1, suggesting groundwater microbial denitrification processes were unlikely in the aquifer. The positive δ15N values coupled with elevated (Fig. 3b) concentrations were more likely the result of soil amendment practices whereby poultry manure is applied to fields during crop replacement cycles to augment soil carbon and nitrogen content (Zebarth et al., 2015). As previously indicated, this site may also have been affected by recent adjacent woodlot clearing and poultry manure application following planting of a new blueberry crop in 2011–2012. If the elevated δ 15N values after January 2012 are omitted from these two wells, the mean δ15N value drops to +4.2 ‰, which corresponds to group 1a. Furthermore, most of the group 1a and 1b wells fall along the same groundwater flow path (Fig. 1).
Wells categorized as group 2 had a mean δ15N of +7.8 ‰, which corresponded to both leachate from manure-treated fields ( ‰; Loo et al., 2017) and poultry manure in general ( ‰). More 15N-depleted samples were likely influenced by synthetic fertilizers or residual soil N, while 15N-enriched samples represented temporal soil zone denitrification. Group 2 wells include 91-03, 91-15, 94Q-14, ABB-02, ABB-03, ABB-05, FT5-12, FT5-25, PB-20, and PB-35. Wells in this group were in the majority, representing 53 % of the sites, and as with group 1 did not exhibit large seasonal or inter-annual swings in concentrations or their δ15N values, other than concentrations and δ15N values that were elevated compared to group 1. Based on these results, it appeared that poultry manure applications or excess residual soil N from historical poultry manure applications influenced the nitrate contamination level in these wells.
The group 3 wells (91-10, US-02, and US-05) had a mean δ15N value of +12.6 ‰ and a δ15N range of +9 ‰ to +16 ‰, which was more 15N enriched than local poultry manure or manure leachates (Table 1). These 15N-enriched results likely resulted from ammonia volatilization of the source poultry manure and temporal soil zone denitrification. Ammonia volatilization occurs in poultry manure piles and during field application. The mineralized residual ammonium can have δ15N values up to +25 ‰ but is dependent on pH, temperature, humidity, and other environmental factors (Kendall, 1998). Group 3 sites are all located down-gradient of current and former poultry barns or known locations of on-field poultry storage piles, which was shown by Wassenaar (1995) to result in 15N-enriched values in soil N from +7.5 ‰ to +13.6 ‰ that are transported to the aquifer.
3.2.2 5-year isotopic trends
The 19 monitoring wells were evaluated based on their nitrate δ15N and δ18O isotopic trends over the 5-year study period (Table 4). The trend evaluation was conducted using Mann–Kendall (monthly data) and Seasonal Kendall (bimonthly data) non-parametric tests for detection of upward or downward trends in a time series at the p > 0.05 level of significance. For individual wells, if there was insufficient evidence to detect a trend, individual well results were grouped as being stable or variable, depending on whether the δ15N standard deviation was < or > 1.0 ‰, respectively. Wells exhibiting seasonality were identified as group B. The analysis showed no statistically significant temporal trend in δ15N during the study period; however, if results from the three nitrate “spiking” sites (US-02, PC-25, and PC-35) were removed, a statistically significant δ15N depletion trend was observed over the 5-year period, with δ15N declining from +9.1 ‰ to +7.4 ‰. This finding corresponded to the previously reported finding of a decadal-scale nitrate 15N depletion trend in the aquifer, which was attributed to a long-term shift from manure to fertilizer use (Wassenaar et al., 2006).
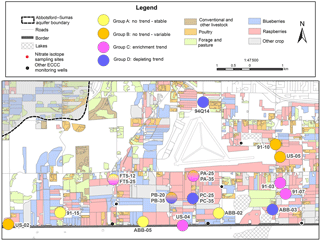
Four wells (91-15, ABB-02, ABB-05, and FT5-12) were classified into trend group A, where analyses did not support a significant upward or downward δ15N trend and ‰ (Fig. 5a). All four wells (21 %) were from distribution group 2, where δ15N values were +6 ‰ to +10 ‰. Interestingly, all group A sites exhibited appreciable variability, but only FT5-12 depicted any seasonality, with peak nitrate concentrations occurring in winter, likely the result of organic soil N mobilization following higher precipitation periods. Average concentrations were 16.1 ± 6.4 mg N L−1. The decoupling of δ15N from suggested a consistent isotopic source, with limited residual build-up of enriched 15N, where concentrations were likely driven by seasonal periods of enhanced recharge.
Trend group B comprised six wells (91-10, PA-25, PA-35, PB-20, US-02, and US-05) with no significant δ15N trend over the study period (Fig. 5b), but exhibiting high δ15N variability around the mean ( ‰). The degree of δ15N and variability differed for most wells in this group; however, all sites exhibited strong δ15N and coupling, with at least a 5 ‰ change in δ15N and 15 mg N L−1 fluctuation in concentrations. Decreasing DO concentrations in US-02 were associated with increasing δ15N values; however, in this case and Cl concentrations (Table S1) were synchronous, suggesting the same source. In fact, the up-gradient field of this well had undergone a renovation cycle in the preceding months, where old raspberry plants were removed followed by a poultry manure soil amendment prior to replanting. Sites 91-10 and US-05 showed similar δ15N and fluctuations, albeit smaller in magnitude, with corresponding increases in chloride and elevated DO concentrations. Sites 91-10 and US-05 are close to each another (< 200 m apart) along a similar groundwater flow path, suggesting these variations are linked. No other sites in this group were spatially proximal. Sites PB-20, PA-25, and PA-35 exhibited varying degree of coupled δ15N and seasonality, suggesting nitrate leaching was the primary driver of variability. For PA-25, increasing concentrations with 15N enrichment (although variable in degree) were systematically observed each winter, suggesting nitrate mobilization occurred during peak winter rainfall periods.
Six sites were identified as trend group C, with increasing δ15N trends (91-03, 91-07, FT5-25, PC-25, PC-35, and US-04). These sites were evenly distributed between distribution groups 1a (3), 1b (2), and 2 (1), suggesting one driver controlling local concentrations and δ15N values. Enriching 15N trends (often along a flow path) are usually associated with progressive microbial denitrification; however, all sites had high DO aerobic concentrations (> 8 mg L−1). Sites PC-25 and PC-35, which exhibited some degree of coupled δ15N and δ18O enrichment at a 2 : 1 ratio, also showed increasing concentrations, suggesting heavy loading of poultry manure. Prior to the marked increase in and δ15N in the spring of 2012, PC-25 and PC-35 exhibited a significant, albeit gradual, increasing 15N trend (Fig. 5c). This revealed a second subtle driver – the increased precipitation that occurred between 2008 and 2011 and its effect on groundwater nitrate concentrations, as shown by Graham et al. (2015). Wells 91-03, FT5-25, and US-04 did not undergo any up-gradient crop replacement or soil amendments and exhibited various degrees of and δ15N seasonality, further strengthening the precipitation link as a potential driver. The δ15N trend could be linked to the enhanced mobilization and infiltration of 15N-depleted organic soil N.
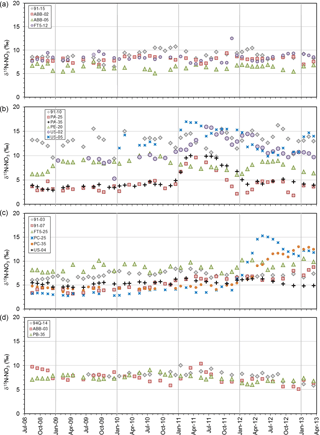
Figure 5δ15N-NO3 time series plots: (a) no trend – stable (SD < ±1.0), (b) no trend – variable (SD > ±1.0), (c) enrichment trend, and (d) depletion trend.
Group D exhibited a 15N depletion trend (Fig. 5d); consisted of monitoring wells 94Q-14, PB-35, and ABB-03; and had a negative δ15N shift of 1 ‰–3 ‰ and δ15N values between +6 ‰ and +10 ‰ (group 2). Well 94Q-14 showed δ15N seasonality, but not in , with concentrations mostly below the MAC. PB-35 showed small seasonality in concentrations but none in δ15N, indicating possible mixing and dilution due to a shift in nitrogen sources. Wassenaar et al. (2006) suggested that a negative δ15N shift may be attributed to the longer-term change in nitrogen sources used from poultry manure to synthetic fertilizers. Lastly, ABB-03 showed no significant trend in concentrations or in δ18O of nitrate; however, δ15N and δ18O were correlated, while δ15N and were inversely correlated. Furthermore, ABB-03 exhibited 3–4-month intervals of lower-than-usual DO conditions (Δ≥4 mg L−1 compared to other seasons; Table S1), which corresponds to periods of 15N enrichment and decreasing NO3, suggesting localized denitrification, that were repeatable to various degrees on a seasonal basis but were most prominent in 2011. These findings suggest localized and temporally limited denitrification may be occurring in the soil root zone in some areas, contributing to 15N enrichment and variability of concentrations. Site ABB-03 was not near Fishtrap Creek (Fig. 1), which Tesoriero (2000) and Wassenaar et al. (2006) identified as a localized riparian denitrification hot spot. Enrichment in 15N of nitrate at these sites appeared to be from temporal drivers that could be overlooked in one-time synoptic sampling (Wassenaar, 1995).
This study represents an unprecedented high-frequency 5-year seasonal spatio-temporal study of the water table well with over 700 nitrate isotopic assays, revealing the dynamics of nitrate recharging the transboundary Abbotsford–Sumas aquifer. The high (monthly) temporal frequency of nitrate and isotopic data aimed to address concerns that infrequent nitrate isotopic or concentration synoptic samplings of shallow groundwater overlook important factors of seasonality that may be key drivers of nitrate sources and fluxes to shallow aquifers. Indeed, our study revealed new important scientific information not previously seen in the synoptic surveys that will help managers better tackle nutrient management strategies to help reduce groundwater pollution.
Overall, and unsurprisingly, we found the predominant perennial source of nitrate to the aquifer at all spatio-temporal scales within the 5-year intensive sampling period was animal waste (poultry) sources, which was already known for decades. Nitrate concentrations in young (< 5 years old) and shallow groundwater were persistently high in nitrate, ranging from 1.3 to 99 mg-N L−1, with a mean of 16.2 mg-N L−1, and well in exceedance of the Canadian drinking water MAC of 10 mg-N L−1 for 76 % of the wells. The study also verified a postulated and subtle decadal-scale shift towards 15N-depleted nitrate sources, likely reflecting systematic changes in agriculture practices from the early days of indiscriminate manure disposal towards more targeted use of synthetic fertilizers or from changes in crop types and associated nutrient practices, as evidenced by the mean δ15N value for nitrate of ‰ compared to ‰ in the 1990s. Synthetic fertilizer and soil N represent a comparatively higher N loading in the central portions of the ASA but are flanked on both sides by higher poultry-manure-dominated N loadings (Fig. 4). The high nitrate concentrations in contemporary recharging groundwater and lack of targeted nitrogen reduction measures means widespread nitrate contamination of the aquifer is likely to persist into the foreseeable future, and our data affirm little evidence for persistent or widespread natural attenuation of nitrate by subsurface denitrification processes at any time of the year. Nitrate remediation of the aquifer will only be possible if agricultural N sources are dramatically reduced or eliminated.
In some wells we found that localized agricultural practices (i.e. nearby organic N soil amendment) had a nearly immediate multi-year negative impact, mainly exhibited by marked increases in poultry-derived N, and lasting for several years and across seasons. This common practice at field renovation and post-woodlot clearing resulted in spatial clustering and differing short-term trends for water table nitrate and its isotopes across the aquifer (Figs. 4 and 6), further revealing that infiltrating and its isotopic composition can change quickly in direct response to contemporary near-field practices. Conversely, this suggests N source cutoff as a groundwater remediation effort could be similarly as effective. Despite 53 % of shallow wells showing no isotopic trends, 47 % showed an 15N enrichment or depletion trend in nitrate, and about half of the wells exhibited nitrate seasonality in concentrations and/or δ15N values controlled by temporal infiltration of residual mineralized N or weak, short-term denitrification.
Due to the rapid shift in and isotopic values of recharging groundwater immediately following field renovation and soil amendment practices, this study reinforces the importance of designing and conducting appropriate spatio-temporal nitrate sampling to reduce the risk of misinterpreting nitrate concentrations and its isotopic data through the more common practice of occasional synoptic surveys. The dynamics of nitrate in younger (< 5 years old) water table wells, however, also imply it would be prudent to monitor deeper, older groundwater which smoothes out short-term fluctuations and hence record longer-term and aquifer-wide trends.
For the ASA agricultural area specifically, measuring the impact of changes in nutrient management practices associated with the switch from raspberry to blueberry crops or field renovation is required to determine its impacts on groundwater nitrate dynamics. Decisions on future groundwater aquifer nitrate management need to take into consideration (i) permanent or cyclical changes in the planned crop types and (ii) the associated nutrient management practices involved with them. Subtle shifts in nitrate in the ASA may be unexpectedly influenced by the recent replacement of raspberries with blueberries, which may to be less reliant of cyclical poultry manure soil amendments.
All data used in this study are available in the Supplement.
The supplement related to this article is available online at: https://doi.org/10.5194/hess-22-4267-2018-supplement.
LIW and BZ prepared the funding proposal. LIW, GG, and MS established the site-selection criteria and selected sites, while MS undertook field activities. MS and LIW analyzed data statistically and conducted the data interpretation. MS prepared the manuscript with the help of LIW, and all co-authors were asked to review the manuscript.
The authors declare that they have no conflict of interest.
Funding for this project was provided by Environment Canada (LIW, MS, GG),
Agriculture and Agri-Food Canada (BZ), and the Canadian Water Network. The
authors would also like to thank Steven Taylor from the Isotope Lab at the
University of Calgary. Geoff Koehler assisted with sampling. Tommy Diep
assisted with GIS mapping and field activities (Environment and Climate
Change Canada).
Edited by: Christine Stumpp
Reviewed by: two anonymous referees
Amberger, A. and Schmidt, H. L.: The natural isotope content of nitrate as an indicator of its origin, Geochim. Cosmochim. Ac., 51, 2699–2705, 1987.
Aravena, R. and Robertson, W.: Use of multiple Isotope Tracers to Evaluate Denitrification in Groundwater, Study of Nitrate from a Large-Flux Septic System Plume, Ground Water, 36, 975–982, 1998.
Aravena, R., Evans, M. L., and Cherry, J. A.: Stable isotopes of oxygen and nitrogen in source identification of NO3 – from septic systems, Ground Water, 31, 180–186, 1993.
Armstrong, J. E., Crandell, D. R., Easterbrook, D. J., and Noble, J. B.: Late Pleistocence stratigraphy and chronology of southwestern British Columbia and northwestern Washington, Geol. Soc. Am. Bull.,76, 321–330, 1965.
Böhlke, J. K.: Groundwater recharge and Agricultural Contamination, Hydrogeol. J., 10, 153–179, 2002.
Böhlke, J. K. and Denver, J. M.: Combined Use of Groundwater Dating, Chemical, and Isotopic Analyses to Resolve the History and Fate of Nitrate Contamination in Two Agricultural Watersheds, Atlantic Coast Plain, Maryland, Water Resour. Res., 9, 2319–2339, 1995.
Canter, L. W.: Nitrates in Groundwater: CRC Press, Lewis Publishing, NY, 1997.
Casciotti, K. L., Sigman, D. M., Hastings, M. G., Bohlke, J. K., and Hilkert, A.: Measurement of the oxygen isotopic composition of nitrate in seawater and freshwater using the denitrifier method, Anal. Chem., 74, 4905–4912, 2002.
Chesnaux, R. and Allen, D. M.: Simulating Nitrate Leaching Profiles in a Highly Permeable Vadose Zone, Environ. Model. Assess., 13, 527–539, https://doi.org/10.1007/s10666-007-9116-4, 2007.
Choi, W. J., Lee, S. M., and Ro, H. M.: Evaluation of Contaminant Sources of Groundwater using Nitrogen Isotope Data: A Review, Geosci. J., 7, 81–87, 2003.
Coplen, T. B.: Reporting of Stable Hydrogen, Carbon, and Oxygen Isotopic Abundances, Pure Appl. Chem., 66, 273–276, 1994.
Cox, S. E. and Kahle, S. C.: Hydrogeology, groundwater quality and sources of nitrate in lowland glacial aquifers of Whatcom county, Washington and British Columbia, Canada, USGS Water Resources Investigation Report, 98–4195, 1999.
Environment Canada: Abbotsford A., Canadian climate normal 1981–2010 station 1100030, available at: http://climate.weather.gc.ca/climate_normals/results_1981_2010_e.html?stnID=702&autofwd=1, last access: 12 October 2014.
Graham, G., Allen, D. M., and Finkbeiner, B.: Climate controls on nitrate concentration variability in the Abbotsford-Sumas aquifer, British Columbia, Canada, Environ. Earth Sci., 73, 2895–2907, 2015.
Halstead, E. C.: Ground Water Supply-Fraser Lowland, British Columbia, Environment Canada, National Hydrology Research Paper, 26, 80 pp., 1965.
Hamilton, P. A. and Helsel, D. R.: Effects of agriculture on groundwater quality in 5 regions of the United States, Ground Water, 33, 217–226, 1995.
Haslauer, C. P., Rudolph, D. L., and Thomson, N. R.: Impacts of changing agricultural land-use practices on municipal groundwater quality: Woodstock, Ontario: Bringing Groundwater Quality Research to the Watershed Scale, 4th International Groundwater Quality Conference, IAHS Publications, 297, 223–233, 2005.
Health Canada: Guidelines for Canadian Drinking Water Quality: Guideline Technical Document – Nitrate and Nitrite, 2013.
Heaton, T. H. E.: Isotopic studies of nitrogen pollution in the hydrosphere and Atmosphere – A Review, Chem. Geol., 59, 87–102, 1986.
Herod, M. N., Suchy, M., Cornett, R. J., Kieser, W. E. Clark, I. D., and Graham, G.: The atmospheric transport of iodine-129 from Fukushima to British Columbia, Canada and its deposition and transport into groundwater, Water Resour. Res., 51, 9628–9645, https://doi.org/10.1002/2015WR017325, 2015.
Hii, B., Liebscher, H., Mazalek, M., and Tuominen, T.: Ground water quality and flow rates in the Abbotsford Aquifer, British Columbia, Environment Canada, Pacific and Yukon Region, 1999.
Kellman, L. M. and Hillaire-Marcel, C.: Evaluation of nitrogen isotopes as indicators of nitrate contamination sources in an agricultural watershed, Agr. Ecosyst. Environ., 95, 87–102, 2003.
Kendall, C.: Tracing nitrogen sources and cycling in catchments, in: Isotope tracers in catchment hydrology, 1st ed., edited by: Kendall C. and McDonnell, J. J., Elsevier Science B.V., Amsterdam, 506, 1998.
Kohut, A. P., Sather, S., Kwong, J., and Chowijka, K.: Nitrate contamination of the Abbotsford aquifer, B.C. Ministry of Water, Land and Air Protection, 18 pp., 1989.
Kowalenko, C. G.: Nitrogen pools and processes in agricultural systems of Coastal British Columbia – A review of publish research, Can. J. Plant Sci. 80, 1–10, 2000.
Kreitler, C. W.: Determining the Source of Nitrate in Groundwater by Nitrogen Isotope Studies: Report of Investigation 83, Bureau of Economic Geology, University of Texas at Austin, Austin Texas, 99, 57, 1975.
Lettenmaier, D. P.: Multivariate Nonparametric-Tests for Trend in Water-Quality, Water Resour. Bull., 24, 505–512, 1988.
Liebscher, H., Hii, B., and McNaughton, D.: Nitrates and pesticides in the Abbotsford aquifer, Southwestern British Columbia, Environment Canada, 83 pp., 1992.
Loo, S. E., Ryan, M. C., Zebarth, B. J., Kuchta, S. H., Neilsen, D., and Mayer, B.: Use of δ15N and δ15O Values for Nitrate source Identification under Irrigated Crops: A Cautionary Vadose Tale, J. Environ. Qual., 46, 528–536, 2017.
Luttmerding, H. A.: Soils of the Langley-Vancouver map area, BC Soil Survey Report No. 15, 1980.
Malekani, F.: A new approach to estimate monthly groundwater nitrate loading from an individual field, MS, University of Calgary, Calgary, Alberta, 2012.
Mariotti, A.: Atmospheric nitrogen is a reliable standard for natural 15N abundance measurements, Nature, 303, 685–687, 1983.
Mitchell, R. J., Babcock, R. S., Gelinas, S., Nanus, L., and Stasney, D. E.: Nitrate Distributions and Source Identification in the Abbotsford-Sumas Aquifer, Northwestern Washington State, J. Environ. Qual., 32, 789–800, 2003.
Ostrom, N. E., Knoke, K. E., Hedin, L. O., Robertson, G. P., and Smucker, A. J. M.: Temporal trends in nitrogen isotope values of nitrate leaching from an agricultural soil, Chem. Geol., 146, 219–227, 1998.
Overdahl, C. J., Rehm, G. W., and Meredith, H. L.: Fertilizer Urea, http://www.extension.umn.edu/distribution/cropsystems/dc0636.html (last access: 10 September 2014), 2007.
Pastén-Zapata, E., Ledesma-Ruiz, R., Harter T., Ramírez, A. I., and Mahlknecht, J.: Assessment of sources and fate of nitrate in shallow groundwater of an agricultural area by using a multi-tracer approach, Sci. Total Environ., 470–471, 855–864, doi.org/10.1016/j.scitotenv.2013.10.043, 2014.
Savard, M. M., Paradis, D., Somers, G., Liao, S., and van Bochove, E.: Winter nitrification contributes to excess in groundwater of an agricultural region, Water Resour. Res., 43, W06422, https://doi.org/10.1029/2006WR005469, 2007.
Savard, M. M., Somers, G., Smirnoff, A., Paradis, D., van Bochove, E., and Liao, S.: Nitrate isotopes unveil distinct seasonal N-sources and the critical role of crop residues in groundwater contamination, J. Hydrol., 381, 134–141, 2010.
Scibek, J. and Allen, D. M.: Modeled Impacts of Predicted Climate Change on Recharge and Groundwater Levels, Water Resour. Res., 42, W11405, https://doi.org/10.1029/2005WR004742, 2006.
Sigman, D. M., Casciotti, K. L., Andreani, M., Barford, C., Galanter, M., and Bohlke, J. K.: A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater, Anal. Chem., 73, 4145–4153, 2001.
Spalding, R. F. and Exner, M. E.: Occurrence of nitrate in groundwater – a review, J. Environ. Qual., 22, 392–402, 1993.
Tesoriero, A. J., Liebscher, H., and Cox, S. E.: Mechanism and rate of denitrification in an agricultural watershed: Electron and mass balance along groundwater flow paths, Water Resour. Res., 36, 1545–1559, 2000.
Thas, O., Van Vooren, L., and Ottoy, J. P.: Nonparametric test performance for trends in water quality with sampling design applications, J. Am. Water Resour. As., 34, 347–357, 1998.
Vorosmarty, C. J., Green, P., Salisbury, J., and Lammers, R. B.: Global water resources: vulnerability from climate change and population growth, Science, 289, 284–288, 2000.
Wassenaar, L. I.: Evaluation of the Origin and Fate of Nitrate in the Abbotsford Aquifer Using the Isotopes of 15N and 18O in NO3, Appl. Geochem., 10, 391–405, 1995.
Wassenaar, L. I., M. Hendry, J. M., and Harrington, N.: Decadal Geochemical and Isotopic Trends for Nitrate in the Transboundary Abbotsford-Sumas Aquifer and Implications for Agricultural Beneficial Management Practices, Environ. Sci. Technol, 40, 4626–4632, 2006.
Xue, D., Botte, J., De Baets, B., Accoe, F., Nestler, A., Taylor, P., Van Cleemput, O., Berglund, M., and Boeckx, P.: Present limitations and future prospects of stable isotope methods for nitrate source identification in surface- and groundwater, Water Res., 43, 1159–1170, 2009.
Zebarth, B. J., Hii, B., Liebscher, H., Chipperfield, K., Paul, J. W., Grove, G., and Szeto, S. Y.: Agricultural land use practices and nitrate concentration in the Abbotsford-Sumas Aquifer, British Columbia, Canada, Agr. Ecosyst. Environ., 69, 99–112, 1998.
Zebarth, B. J., Ryan, M. C, Graham, G., Forge, T. A., and Neilsen, D.: Groundwater Monitoring to Support Development of BMPs for Groundwater Protection: The Abbotsford-Sumas Aquifer Case Study, Ground Water Monit. R., 35, 82–96, https://doi.org/10.1111/gwmr.12092, 2015.