the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Technical note: Efficiency of various evaporation barriers for use in automated water samplers for subsequent water isotope analysis
Christin Mueller
Tim Giorgio Pekarev
Kay Knoeller
High temporal and spatial resolution water sampling of stream water or precipitation for subsequent water stable isotope analysis (δ2H and δ18O) is commonly conducted with self-made or commercial automated samplers. However, prolonged storage of water samples in open bottles inside autosamplers can lead to isotopic alterations caused by evaporative fractionation and diffusion driven by vapor pressure gradients.
In this study, we tested the effectiveness of different evaporation barriers – tube-dip-in, ball valves, and siphons – under controlled laboratory conditions. Experiments were conducted using glass bottles with varying filling levels (50 to 250 mL in 250 mL bottles) and exposure times of up to 52 d. Our results demonstrate that all tested barriers effectively reduce isotopic alterations. Among these, the tube-dip-in emerged as the simplest and most efficient solution, reliably preserving isotopic data even over extended storage durations. We also observed isotopic differences between the water in the tube-dip-in and the main bottle. This phenomenon becomes particularly relevant when sample volumes are very low, as water from the tube-dip-in may influence the overall isotopic composition of the sample. This finding highlights the need for careful consideration of bottle design and sample handling, especially with low water volumes.
These findings provide valuable insights for the design of cost-effective, automated water sampling systems for stable isotope applications, emphasizing the importance of evaporation barriers to ensure reliable and accurate isotopic analyses.
- Article
(4165 KB) - Full-text XML
-
Supplement
(386 KB) - BibTeX
- EndNote
Isotope analysis of water requires collecting and storing samples without long-term and large-scale interference to the gas phase to ensure results are as close as possible to true values. While this may sound straightforward, it presents significant challenges, as sampling often introduces the largest errors in analytical outcomes (Esbensen, 2020; Romañach et al., 2021). Therefore, it is crucial to focus on refining sampling techniques. Numerous sampling systems for water collection are available, each suited to specific applications or climatic conditions. In the publication from Michelsen et al. (2018) various collectors used in isotope hydrology are compared, while recent works (von Freyberg et al., 2020; Carton et al., 2024a; Carton et al., 2025) modified existing samplers.
Cavity Ring-Down Spectroscopy (CRDS) exemplifies how the growing accessibility and efficiency of laser spectrometry have significantly reduced the cost of isotopic measurements while providing a user-friendly, and relatively precise method in analyzing stable isotope ratios in water molecules (Hachgenei et al., 2022). This advancement allows for an increased number of samples to be analyzed rapidly, facilitating better understanding of the water cycle and associated processes. A critical prerequisite for successful rainwater sampling is the strict prevention of evaporation during the collection and storage period, as even minimal losses can lead to a significant enrichment of heavy isotope in the water phase and compromise data quality (Schürch et al., 2003).
Extreme events often catch researchers unprepared, forcing them to use improvised sampling containers like empty soda bottles or honey jars (Fischer et al., 2019). Financial constraints may also limit access to commercial bottles or automated sampling devices. To address these challenges, do-it-yourself (DIY) instructions for constructing a rainwater sampler have become increasingly common in the field of isotope hydrology (e.g. Prechsl et al., 2014; Fischer et al., 2019; von Freyberg et al., 2020; Carton et al., 2024b). They explored different low-tech, low-budget rainfall sampler for stable isotope analysis, assessing there effectiveness in maintaining sample quality. Such innovations highlight the continuous efforts to improve sampling systems, making them more efficient, reliable, and compatible with modern analytical requirements.
Among commercially available systems, the widely used cumulative rain water sampler (RS1) for water isotope analysis first published by Gröning et al. (2012) and sold by Palmex Ltd. employs a 3 L bottle with a long tube (15 m polypropylene plastic hose with an inner diameter of 5 mm) to prevent evaporation. This system forms the backbone of the International Atomic Energy Agencys' Global Network of Isotopes in Precipitation (GNIP) (IAEA, 2014), which continues to expand (Terzer-Wassmuth et al., 2021, 2022) (Link website: https://nucleus.iaea.org/wiser/, last access: 17 January 2025). The International Atomic Energy Agency (IAEA) has also developed detailed technical guidelines for sampling procedures, ensuring consistency across its global network of stations (Gat, 1981).
Early solutions to evaporation challenges included the use of paraffin oil, which forms a protective layer over collected rainwater. In such designs, a 5 mm-thick oil layer prevents evaporation during the collection period (WMO, 1972). However, these systems present practical difficulties as it can be challenging to remove the oil from the water as it can form a gelatinous phase and leave behind residues that can contaminate laboratory equipment such as mass spectrometers (Friedman et al., 1992; Gröning et al., 2012). An alternative is described by Weaver and Talma (2007) who created a cumulative rainfall sampler using a tennis ball and oil for water and chloride isotope studies. Michelsen et al. (2018) compared precipitation collectors commonly used in isotope hydrology, providing an overview of various sampling systems and the challenges associated with their use (WMO, 1972).
High-resolution sampling is essential for many scientific investigations, as monthly intervals are often inadequate for addressing research questions that require detailed temporal data. Numerous studies have shown that the isotopic composition of precipitation can vary significantly during individual rainfall events (e.g. McDonnell et al., 1990; Munksgaard et al., 2012; Aemisegger et al., 2015; Fischer et al., 2017; Graf et al., 2019). Capturing these variations in stable isotope composition requires sequential sampling with high temporal resolution. Applications such as numerical models (e.g., transSAS from Benettin and Bertuzzo (2018) and used in Radtke et al. (2024)) particularly benefit from high-resolution, volume-based sampling. Additionally, understanding water residence times demands monitoring multiple compartments, such as precipitation and surface water, across various locations within a catchment.
Automatic samplers, essential for collecting high-frequency data, often cannot be emptied immediately after extreme events due to the inaccessibility of study areas. This limitation may lead to extended storage times, which can result in detectable isotope enrichment (Michelsen et al., 2018; Nigro et al., 2024). These challenges highlight the need for evaporation-proof sampling systems.
All these sampling systems aim to minimize or prevent the enrichment of heavy isotopes in the water phase due to evaporation. For both scientific research and commercial applications, laboratory tests should be conducted prior to their use in scientific projects or before market release to validate the suitability and operational range of the devices. In response to the need of a low-cost, autonomous water sampler tailored for isotopic water analysis, we conducted this experimental approach, which is of significant interest to the scientific community. This study presents laboratory tests evaluating potential evaporation barriers, focusing on factors such as bottle filling and exposure time. Our goal is to create a robust and adaptable system that can be easily used by a large number of users. The findings provide valuable insights into sampling system design and identify potential challenges associated with sampling systems deployed over extended periods.
2.1 Design and Setup
The experiments aimed to identify an effective evaporation barrier for water samples intended for isotopic analysis. Generally, SCHOTT glass bottles were used to store the water samples during the experiment. For the tubing, polypropylene hoses were used with an inner diameter of 5 mm. As evaporation barrier, three inlet designs were tested (Fig. 1): (1) inserting a tube before the sampling bottle to act as a siphon; (2) using a tube-dip-in, which extends into the sampling bottle to minimize direct exposure to the atmosphere; and (3) employing a ball valve that opens when the bottle is filling and closes when the water flow stops, using a plastic ball to seal the opening. Due to the functionality of the ball valve, it can only be used in combination with a siphon because it needs to be filled from the bottom to allow the ball to float and open the valve.
The experimental procedures were divided into two setups. In the first setup, all bottle inlets were equipped with siphons as evaporation barriers. In the second setup, no siphons were used. The designs and specific configurations of each method are explained in more detail below.
In the first experiment, we were testing different evaporation barriers at the filling side of a glass sampling bottle which were combined or isolated used from each other: we tested tube-dip-in, ball-valve and a siphon. The design of the test bottles (n = 44) varied at the inlet, with some equipped with a tube-dip-in and/or a ball valve with siphon, while others had neither (Fig. 1a). The bottles with siphons were filled through the siphon, ensuring that a water column remained inside as a barrier. In bottles with a ball valve, the valve was positioned along the filling tube and after the siphon. The principle of the ball-valve is a plastic ball which starts to float as water entered the bottle and then sink back into place to seal the inlet after filling. To test only the effectiveness of the inlet system, the outlet vent of all bottles was sealed with a stopper, preventing any influence from evaporation through the outlet. The water volumes added to the bottles were 50, 100, 150, 200, or 250 mL, with exposure durations of 5, 7, 20, or 21 d at a room temperature of approximately 23 °C. After the experiment, water from the bottle and the siphon was sampled for subsequent water isotope analysis. Additionally, reference bottles – both sealed (n=38) and open (n=14) – were subjected to the same variation in filling volume and exposure time and were also sampled for water isotope analysis.
Based on the results of experiment 1, we selected the evaporation barrier that was easiest to handle and demonstrated the most reliable performance compared to the other tested evaporation barriers. The second experiment focused on a long-term assessment of this evaporation barrier. For this, we decided to use the tube-dip-in system combined with an extended tubing setup consisting of a 15 m long PP tube with an inner diameter of 5 mm (Fig. 1b). This setup, first mentioned by Gröning et al. (2012) allows for the release of air during the filling process but also serves as an effective evaporation barrier during storage. In Carton et al. (2024b) calculations of the tubing length were performed based on temperature and pressure variations, applying the ideal gas law. The study concluded that a tubing length of 4 m is sufficient for 125 mL bottles in a monthly sampling.
All tests were conducted under controlled laboratory conditions, with a mean temperature of 23 °C and a relative humidity of 50 %. No variations in temperature or humidity were forced, which could have further increased evaporation. Sampling was performed on both tube-dip-in water and bottled water (BW), followed by water isotope analysis (n=19). The sampling of the tube-dip-in water was performed using a straw-like principle, where the water inside the tube was held by blocking one end of the tube and releasing the water by removing the finger from the other end. The exposure times in this experiment were 12, 26, 39, or 52 days, and tests were conducted with two different sample volumes (50 mL or 250 mL). All data are summarised in Table S2 (Supplement). Open and closed reference bottles (n = 10 for each) were handled with similar variations in filling volume and exposure time.
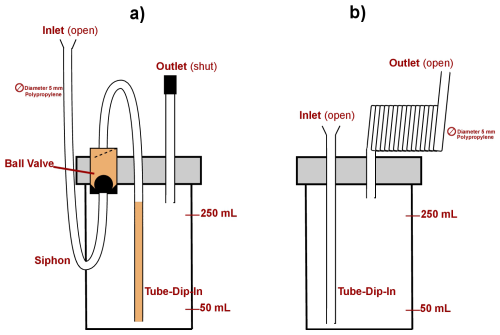
Figure 1(a) Experimental set-up of the first experiment, which used siphon, ball-valve, and tube-dip-in as evaporation barriers. Light orange parts (ball-valve and tube-dip-in) could be removed or added. (b) Experimental set-up of the second experiment, which used a tube-dip-in as evaporation barrier. Here the outlet was a 15 m long open tube with an inner diameter of 5 mm. The type of bottle (SCHOTT glass bottle) used remained unchanged throughout the experiment.
2.2 Laboratory methods
A liquid isotope analyzer (Picarro L2120-i) was utilized to conduct duplicate measurements of stable isotopic signatures in water. For this purpose, 20 replicates of internal standards, which had been calibrated to Vienna Standard Mean Ocean Water (VSMOW) and Standard Light Antarctic Precipitation (SLAP) certified reference materials, were analyzed to normalize the samples to the VSMOW-SLAP scale. The analytical uncertainties were determined to be ±0.15 ‰ for δ18O and ± 0.6 ‰ for δ2H. The isotopic ratios are reported in delta notation relative to VSMOW for both the oxygen and hydrogen isotope signatures of water.
2.3 Statistics
The statistical analysis was conducted with Python 3.9.7. Data were grouped by experimental setups: experiment 1 (siphon) and experiment 2 (no siphon). The variables weight loss, δ2H, δ18O and deuterium excess were tested for variance homogeneity for each experiment using the Levene test, followed by a Shapiro-Wilks-Test to test for normal data distribution in the groups. Homogeneity of variance was not established, and normality was limited to only a few groups. Therefore, and due to the small sample size, the Kruskal-Wallis test was applied to assess differences between the groups, followed by Dunn's post hoc test with Bonferroni correction. The null hypothesis assumed no significant differences in weight loss or isotopic composition changes among the tested closure systems.
3.1 Weight loss due to evaporation
No weight loss was observed in the closed bottles during the experiments, regardless of fill volume or exposure time (Mean = 0.03 %, n = 26, SD = 0.05 %) (Fig. 3). The highest relative weight loss was observed in the open bottles, which served as a reference. Regarding the efficiency of the tested evaporation barriers, the lowest volume loss was recorded with the tube-dip-in system and siphon (Mean = 0.12 %, n = 4, SD = 0.09 %). However, the sample size in the first experimental setup was limited. Consequently, the experiment only with a tube-dip-in system was intensified in the second setup, focusing exclusively on this method to further validate the results. Very low volume losses were observed when both the tube-dip-in system and a ball valve were used in combination with a siphon (Mean = 0.61 %, n = 12, SD = 1.55 %). When the setup included a ball valve in combination with a siphon but without a tube-dip-in, weight loss increased slightly (Mean = 1.14 %, n = 8, SD = 1.78 %). The siphon alone, without the ball valve or tube-dip-in, showed the highest evaporation loss compared to all tested barriers (Mean = 1.38 %, n = 10, SD = 1.3 %). This method also posed a risk of sample compromising, as temperature or pressure differences (even under laboratory conditions) caused water movement within the siphon. Significant weight loss was observed when the siphon emptied completely, rendering the barrier inactive.
The duration of the experiment (comparing two groups: 5 and 7 d vs. 20 and 21 d) showed no significant impact on evaporation loss. However, the amount of sample in the bottle had a significant effect on percentage weight loss, as expected.
Different significance levels (e.g., p<0.05, p<0.01, p<0.001) indicate the likelihood that an observed result was due to chance under the null hypothesis. Smaller p-values correspond to lower probabilities of random occurrence and provide stronger evidence against the null hypothesis. In Fig. 3, the highest significance (p<0.001) is observed for the references (open vs. closed) as well as for the closed bottle compared to the siphon-only setup in the experiment. Statistical significance (p<0.05) for evaporation is still evident in the experimental setup where only a siphon and a ball valve were used.
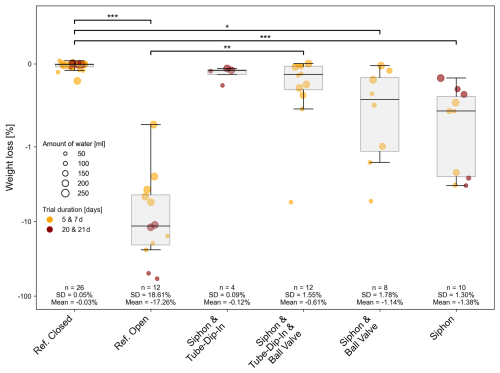
Figure 3Weight difference between the start and end of experiment 1 in % was used as an indicator of evaporation on a logarithmic scale. The exposure time ranged from short-term (5 to 7 d) to long-term (20 to 21 d). The initial water volumes in the 250 mL SCHOTT glass bottles were 50, 100, 150, 200, or 250 mL. Various evaporation barriers were tested, with a closed and open bottle serving as the references. Significant differences between groups, determined via Dunn's post-hoc test, are indicated by horizontal lines with corresponding significance levels p < 0.05 (*), p < 0.01 (), p < 0.001 ().
3.2 Deuterium excess as indicator for evaporation efficiency
Deuterium excess refers to the deviation of water isotope data in permill from the GMWL in dual isotope space, as defined by the Dansgaard's equation: d-excess =δ2H − 8 ⋅ δ18O (Dansgaard, 1964). It provides valuable insights into the processes affecting water's isotopic composition. This second-order isotope parameter is particularly useful for identifying the effects of evaporation and water isotopic fractionation. Deviations from the GMWL typically indicate isotopic fractionation caused by physical processes, such as evaporation. Isotope data plotting on the GMWL have a deuterium excess value of 10 ‰, suggesting no significant fractionation or evaporation, which reflects natural isotopic ratios typical of rainwater (Craig, 1961; Dansgaard, 1964). The GMWL as reference for deuterium excess values is shown in Fig. 4, along with the measurement accuracy (±1.34 ‰) in grey. Deuterium excess values from experiment 1 indicate the highest significance (p < 0.001) for the reference water compared to the siphon water. In general, the siphon water exhibits the lowest deuterium excess values, indicating the highest extent of evaporation (siphon water after 21 d, d-excess = 3.62 ‰, see Table S1 (sample ID 103) in the Supplement). All other evaporation barriers (tube-dip-in, ball-valve in combination or alone) show variations within the measurement accuracy. The duration of storage and amount of sample, indicated by the colour and symbol size, respectively, do not have a significant impact in the experiment (Fig. 4).
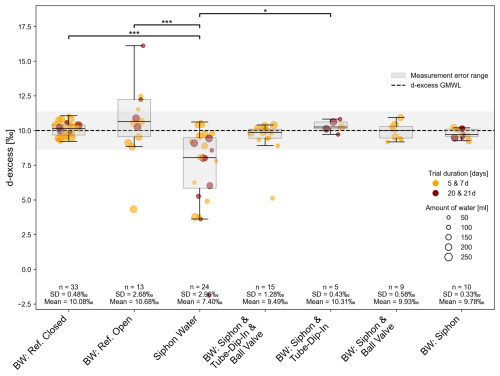
Figure 4Deuterium excess from experiment 1 as an indicator of isotope fractionation on H2O through different time steps within the evaporation experiment. Significant differences between groups, determined via Dunn's post-hoc test, are indicated by horizontal lines with corresponding significance levels p < 0.05 (*), p < 0.01 (), p < 0.001 ().
3.3 Water isotopic signatures in evaporation experiments using various barriers
Results of water isotope measurements from the first evaporation experiment using different evaporation barriers are shown in Fig. 5. The closed reference bottle provides the water isotope signature used as the target reference (δ2H = −57.6 ± 0.7 ‰ and δ18O = −8.5 ± 0.1 ‰) and serves as a control throughout the experiment. The open reference bottle is clearly unsuitable and exhibits the highest water isotope shifts (mean δ2H = −44.5 ± 16.7 ‰, mean δ18O = −6.9 ± 1.8 ‰), which are significant.
Throughout the duration of the experiment, all tested evaporation barriers yielded reliable results. The tube-dip-in system, ball valve, and siphon showed no significant differences among their results of water isotopes (Fig. 5). However, when a siphon is used as a barrier, it must be ensured that the water standing in the siphon does not mix with the sample, as its isotopic signature shows significant enrichment. Notably, if the sample volume is large, the impact of potential contamination from siphon water becomes less pronounced. It should be noted that this experiment was conducted under laboratory conditions with minimal variation in temperature and humidity. Further testing under variable environmental conditions is recommended, particularly for evaporation barriers employed in long-term studies and in regions subject to significant temperature and humidity fluctuations.
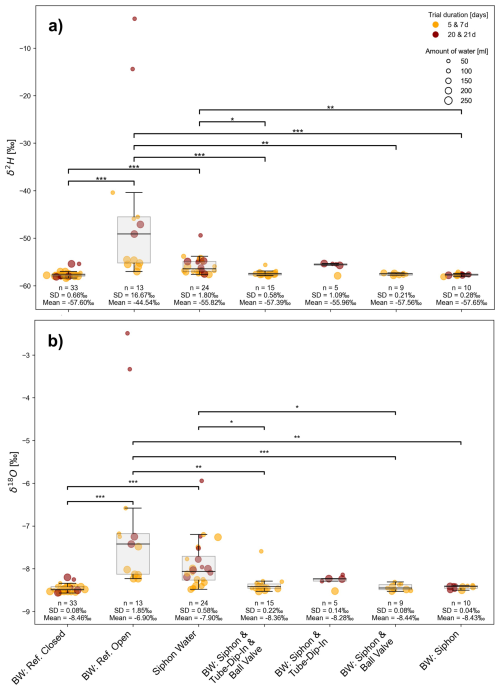
Figure 5δ2H (a) and δ18O (b) isotopic signature [‰] from experiment 1, indicating isotope fractionation of H2O over different time steps in the evaporation experiment. Significant differences between groups, determined via Dunn's post-hoc test, are indicated by horizontal lines with corresponding significance levels, p < 0.05 (*), p < 0.01 (), p < 0.001 ().
3.4 Long-Term experiments with tube-dip-in as evaporation barrier: Findings from weight loss and water isotope analysis
In experiment 2, the weight loss was measured after four exposure periods (13, 26, 39, or 52 d) for two different filling volumes (50 mL or 250 mL). The efficiency of the tube dip-in system was tested under laboratory conditions. Closed and open bottles were used as references and ran throughout the entire experiment. Figure 6 clearly shows on a logarithmic scale that open reference bottles experienced a relative weight loss of up to 100 %. The average relative weight loss was 40 %. In general, the lower the filling volume and the longer the exposure time, the higher the percentage of relative weight loss. The relative weight loss in closed bottles was negligibly small. The relative weight loss with the tube-dip-in system as the evaporation barrier averaged 0.31 %. The maximum relative weight loss was less than 1 %. The greatest amount of relative weight loss occurred with lower filling volumes.
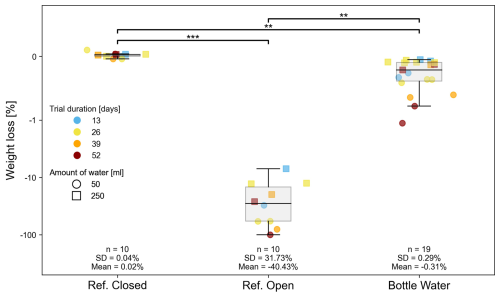
Figure 6The relative weight difference between the start and end of experiment 2, expressed as a percentage on a logarithmic scale, was used as an indicator of evaporation. The exposure time ranged from 13 to 52 d. The initial water volumes in the 250 mL SCHOTT glass bottles were 50 or 250 mL. The tested evaporation barrier, the tube-dip-in system, is shown in relation to closed and open bottles serving as references. Significant differences between groups, determined via Dunn's post-hoc test, are indicated by horizontal lines with corresponding significance levels p < 0.05 (*), p < 0.01 (), p < 0.001 ().
Deuterium excess data are summarized in Fig. 7. For the long-term experiment, we measured the isotopic signature of a standard water before, during, and after the experiment. The d-excess values remained within the measurement accuracy (mean 10.4 ± 0.5 ‰). The closed reference bottle exhibited a similar variation (10.1 ± 0.5 ‰), confirming the proper storage conditions of the standard water and the seal integrity of the bottles used. The lowest d-excess was observed in water from the tube-dip-in (min −11.8 ‰), with its isotopic signature significantly different from the closed system (p < 0.001). Throughout the experiment, the bottled water displayed d-excess values within the measurement error range (mean 9.43 ± 0.53 ‰). Only d-excess values of samples with a standing time of 52 days deviated from the GMWL by more than the measurement uncertainty, showing significant evaporation effects (minimum d-excess of 8.4 ‰). The reason why the water samples taken from the open reference bottle plot along a line with a slope close to 8 remains unclear. One possible explanation is a higher relative humidity in the open headspace above the water surface and/or fluctuations in relative humidity and temperature during the experiment. These factors may have induced interactions between evaporation and condensation processes. We only observe this effect in open-bottle experiments because the interface between water and the open laboratory atmosphere is significantly larger than in all other setups of our experimental series. However, the highest variations in water isotope signatures (δ2H δ18O – H2O) were observed for the open reference bottle (Fig. 8), with mean values of −25.4 ± 27.5 ‰ for δ2H and −4.4 ± 3.4 ‰ for δ18O in the water. The fluctuations in water isotopes in the tube-dip-in were much smaller (mean δ2H = −51.8 ± 2.9 ‰, mean δ18O = −6.7 ± 1.1 ‰), yet still significantly different (p = 0.001) from the water sample stored in a closed bottle. The sample water in the bottle (BW) showed no evaporation effects throughout the experiment duration (max. 52 d). No significant differences in water isotope values were observed between BW and the closed reference bottle. Additionally, the sample volume appears to play a minor role. However, for the water in the tube-dip-in, differences between sample volumes were noticeable. The smaller the sample volume in the bottle, the greater the evaporation effects observed in the tube-dip-in water.
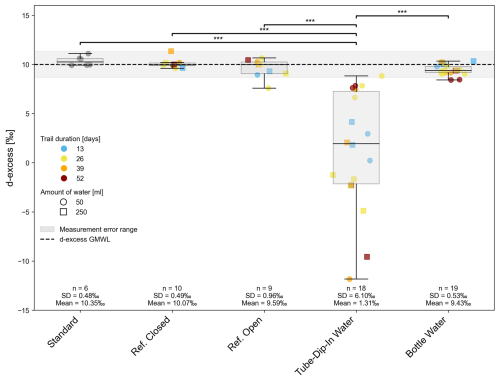
Figure 7Deuterium excess from experiment 2 as an indicator of isotope fractionation on H2O through different time steps within the evaporation experiment. Significant differences between groups, determined via Dunn's post-hoc test, are indicated by horizontal lines with corresponding significance levels p < 0.05 (*), p < 0.01 (), p < 0.001 ().
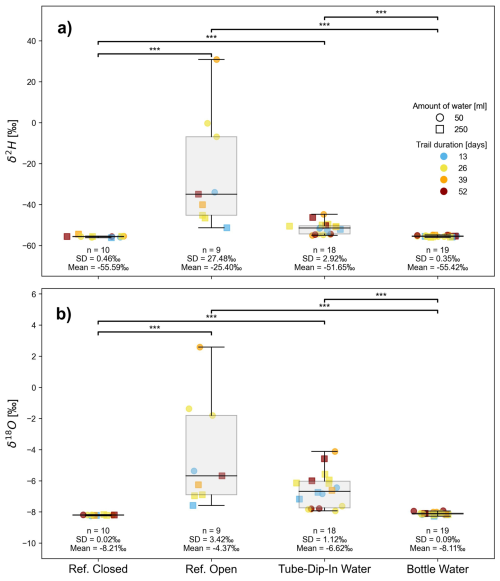
Figure 8δ2H (a) and δ18O (b) isotopic signature [‰] from experiment 2 as an indicator of isotope fractionation on H2O through different time steps within the evaporation experiment. Significant differences between groups, determined via Dunn's post-hoc test, are indicated by horizontal lines with corresponding significance levels p < 0.05 (*), p < 0.01 (), p < 0.001 ().
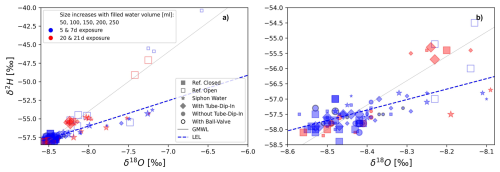
Figure 9Dual isotope plot showing isotope fractionation during experiment 1 using different evaporation barriers (siphon, ball valve, and tube-dip-in). Both diagrams (a, b) display the same results on different scales. The label sizes correspond to the filled water volume, which varies between 50, 100, 150, 200, or 250 mL. Colors represent different exposure times, with blue indicating short exposure and red indicating long exposure. The equation of the local evaporation line (LEL) is δ2H = 3.4 ⋅ δ18O − 28.6 with R2 = 0.52 and p-value < 0.001.
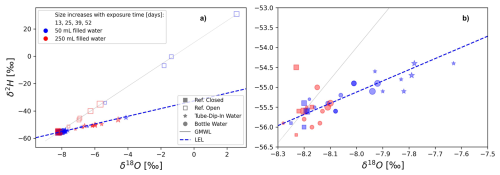
Figure 10Dual isotope plot showing isotope fractionation during experiment 2 using tube-dip-in as the evaporation barrier. Both diagrams (a, b) display the same results on different scales. Label sizes correspond to the exposure time, which varied between 13, 25, 39 and 52 d. Colors represent different water volumes (50 mL vs. 250 mL). The star labels indicate water samples from the tube-dip-in. The equation of the local evaporation line (LEL) is δ2H = 2.8 ⋅ δ18O − 32.6, R2 = 0.52, p-value < 0.001.
3.5 Dual isotope plots for different experimental investigations
The following section evaluates the dual isotope plots for both experiments. Figure 9 illustrates the results of the first experiment, which used multiple evaporation barriers. Both diagrams (a and b) display the same data, but Fig. 9a provides an overview, while Fig. 9b focuses on the details. The isotope values of the closed reference bottle plot along the Global Meteoric Water Line (GMWL) with minimal variability. In contrast, the isotope values of the water sample stored in the open reference bottle exhibit greater variability. The isotopic values of water sampled using the siphon (labeled with a star) plot along an evaporation line with a lower slope than the GMWL. Here, exposure times appear to play a minor role. Similarly, for water samples in bottles with different evaporation barriers, exposure time also seems to be of lesser importance. The dual isotope plot of the long-term experiment using the tube-dip-in is shown in Fig. 10. Here, much more significant is the filling volume of the sample bottles, which is represented by symbol size. The smaller the filling volume, the larger the deviation from the GMWL, indicating greater evaporation. Similar findings were reported by Carton et al. (2024b) and Natali et al. (2024). This underscores that the choice of an appropriate bottle size is of high importance for maintaining the stability of a water isotope signature of water samples stored with a gas phase. The bottle size should align with the expected volume of collected water (e.g., rainfall), considering the funnel area and desired sample volume. Overflow should be avoided to prevent water loss, which could compromise subsequent analyses and hydrological interpretations.
The isotopic signature of the water within the tube-dip-in exhibits significant isotopic fractionation. The results plot along an evaporation line (indicated by star-shaped symbols). In the long-term experiment, fractionation effects become evident with prolonged exposure times, as reflected by the label sizes.
For short exposure times (13, 26 d), the variation remains within the measurement accuracy (Fig. 10b). As exposure time increases, a trend of enrichment in the water sample becomes apparent. However, the variation of all samples with certain evaporation barriers (mean δ2H = −53.5 ± 6.2 ‰, mean δ18O = −8.1 ± 0.1 ‰) remains minimal. A significant enrichment is observed in the tube-dip-in water (variation up to −44.8 ‰ for δ2H and −4.4 ‰ for δ18O). Some experimental bottles show less pronounced enrichment compared to others. Generally, the trend of higher enrichment with longer exposure times is evident.
In this study, we investigated various evaporation barriers to reduce evaporative enrichment and diffusion driven by vapor pressure gradients in water samples collected for subsequent water stable isotope analysis. Laboratory experiments were conducted using different barrier setups, fillings, and exposure times. Our findings demonstrate that water samples with certain evaporation barriers show significantly less isotopic alteration due to evaporative enrichment and diffusion driven by vapor pressure gradients compared to samples stored in open bottles. The simplest and most effective setup tested was a combined tube-dip-in system and a long tubing to release air during filling. This configuration allowed gases to escape during filling while compensating pressure and temperature-induced volume changes during the observation period. The tube-dip-in system is characterized by its simplicity, ease of application, and low susceptibility to failure. These features make it particularly suitable for long-term evaporation experiments. This method proved to be a practical and efficient evaporation barrier, achieving isotopic fractionation levels comparable to those observed in closed systems. However, significant isotopic differences were noted between the water in the tube-dip-in and the water in the main bottle. These differences are likely to be more evident under variable temperature and humidity conditions. We recommend removing the tube-dip-in water by maintaining the pressure in the tube while opening the bottle (similar to the “straw principle”) and then discarding the tube-dip-in water. To ensure the effectiveness of this approach, evaporation and condensation in the bottle must be minimized so that the more evaporated water remains in the tube-dip-in system. In this study, temperature and relative humidity were kept relatively constant under laboratory conditions. However, future experiments should systematically vary and control these parameters to better understand their extent of interactions in driving isotopic changes.
Alternative evaporation barriers, such as ball-valves or siphons, can also be used. However, these methods come with higher maintenance requirements. Ball-valves may require regular cleaning or replacement and are prone to clogging, while siphons are susceptible to sample contamination if emptied due to large temperature or pressure fluctuations. Such incidents can create open conditions that accelerate evaporation.
Overall, our results highlight the importance of selecting evaporation barriers that are tailored to specific sampling conditions and maintenance requirements. For the tested scenarios, the “tube-dip-in only” setup proved to be the simplest and most effective solution for preserving the isotopic integrity of water samples. However, in cases where environmental conditions (temperature and humidity) fluctuate significantly, more complex systems, such as a combination with a siphon, may be considered. These methods provide additional control over evaporation but require higher maintenance and pose a risk of sample contamination under weather-related pressure fluctuations. If samples are collected over extended periods rather than as discrete events, siphon systems are not suitable. In such cases, siphon water may repeatedly mix with the sample, leading to potential contamination and altering the isotopic composition due to cumulative evaporation effects.
The water stable isotope measurements from the two experiments are provided as Supplement.
The supplement related to this article is available online at https://doi.org/10.5194/hess-30-1801-2026-supplement.
CM: Conceptualization, experimental design, manuscript writing, funding acquisition, supervision, and project administration; TGP: Laboratory experiments, statistical analysis, Python scripting, review, and editing; KK: Conceptualization, resources, review, and editing.
The contact author has declared that none of the authors has any competing interests.
Publisher’s note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. While Copernicus Publications makes every effort to include appropriate place names, the final responsibility lies with the authors. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This work was supported by UFZ's innovation program transfun®, with special thanks to Joachim Noeller and Lydia Woiterski for their continued support. We would also like to express our gratitude to Karl-Heinz Walz and Manuel Schädler from MAXX Mess- und Probenahmetechnik GmbH for their valuable discussions and for providing the various evaporation barriers used in our laboratory testing. Their contributions were crucial to the success of this study. Additionally, we thank the IAEA for the opportunity to participate in CPR F31007, where the topic of isotope sampling systems was discussed in depth.
This research has been supported by the Helmholtz-Gemeinschaft (UFZ innovation program transfun®).
The article processing charges for this open-access publication were covered by the Helmholtz Centre for Environmental Research – UFZ.
This paper was edited by Natalie Orlowski and reviewed by three anonymous referees.
Aemisegger, F., Spiegel, J. K., Pfahl, S., Sodemann, H., Eugster, W., and Wernli, H.: Isotope meteorology of cold front passages: A case study combining observations and modeling, Geophysical Research Letters, 42, 5652–5660, https://doi.org/10.1002/2015GL063988, 2015.
Benettin, P. and Bertuzzo, E.: tran-SAS v1.0: a numerical model to compute catchment-scale hydrologic transport using StorAge Selection functions, Geosci. Model Dev., 11, 1627–1639, https://doi.org/10.5194/gmd-11-1627-2018, 2018.
Carton, C., Barbecot, F., Hélie, J.-F., Horoi, V., and Birks, S.: Adapting automatic water samplers for the isotopic study of rainfall at high temporal resolution, Authorea [preprint], https://doi.org/10.22541/au.172537935.54322578/v1, 2024a.
Carton, C., Barbecot, F., Hélie, J.-F., Horoi, V., Birks, J., Picard, A., and Mona, J.: Affordable event and monthly rain samplers: Improving isotopic datasets to understand meteorological processes, Rapid Communications in Mass Spectrometry, 38, e9710, https://doi.org/10.1002/rcm.9710, 2024b.
Carton, C., Barbecot, F., Hélie, J.-F., Horoi, V., and Birks, J.: Adapting Automatic Water Samplers for the Isotopic Study of Rainfall at High Temporal Resolution, Rapid Communications in Mass Spectrometry, 39, e10017, https://doi.org/10.1002/rcm.10017, 2025.
Craig, H.: Isotopic Variations in Meteoric Waters, Science, 133, 1702–1703, https://doi.org/10.1126/science.133.3465.1702, 1961.
Dansgaard, W.: Stable isotopes in precipitation, Tellus, 16, 436–468, https://doi.org/10.1111/j.2153-3490.1964.tb00181.x, 1964.
Esbensen, K.: Introduction to the Theory and Practice of Sampling, https://doi.org/10.1255/978-1-906715-29-8, 2020.
Fischer, B. M. C., van Meerveld, H. J., and Seibert, J.: Spatial variability in the isotopic composition of rainfall in a small headwater catchment and its effect on hydrograph separation, Journal of Hydrology, 547, 755–769, https://doi.org/10.1016/j.jhydrol.2017.01.045, 2017.
Fischer, B. M. C., Aemisegger, F., Graf, P., Sodemann, H., and Seibert, J.: Assessing the Sampling Quality of a Low-Tech Low-Budget Volume-Based Rainfall Sampler for Stable Isotope Analysis, Frontiers in Earth Science, 7, https://doi.org/10.3389/feart.2019.00244, 2019.
Friedman, I., Smith, G. I., Gleason, J. D., Warden, A., and Harris, J. M.: Stable isotope composition of waters in southeastern California 1. Modern precipitation, Journal of Geophysical Research: Atmospheres, 97, 5795–5812, https://doi.org/10.1029/92JD00184, 1992.
Gat, J. R.: Stable isotope hydrology Deuterium and oxygen-18 in the water cycle, IAEA, International Atomic Energy Agency (IAEA), ISBN 92-0-145281-0, 1981.
Graf, P., Wernli, H., Pfahl, S., and Sodemann, H.: A new interpretative framework for below-cloud effects on stable water isotopes in vapour and rain, Atmos. Chem. Phys., 19, 747–765, https://doi.org/10.5194/acp-19-747-2019, 2019.
Gröning, M., Lutz, H. O., Roller-Lutz, Z., Kralik, M., Gourcy, L., and Pöltenstein, L.: A simple rain collector preventing water re-evaporation dedicated for δ18O and δ2H analysis of cumulative precipitation samples, Journal of Hydrology, 448–449, 195–200, https://doi.org/10.1016/j.jhydrol.2012.04.041, 2012.
Hachgenei, N., Vaury, V., Nord, G., Spadini, L., and Duwig, C.: Faster and more precise isotopic water analysis of discrete samples by predicting the repetitions' asymptote instead of averaging last values, MethodsX, 9, 101656, https://doi.org/10.1016/j.mex.2022.101656, 2022.
IAEA: GNIP precipitation sampling guide, IAEA Water Resources Program, 2, 4–15, 2014.
McDonnell, J. J., Bonell, M., Stewart, M. K., and Pearce, A. J.: Deuterium variations in storm rainfall: Implications for stream hydrograph separation, Water Resources Research, 26, 455–458, https://doi.org/10.1029/WR026i003p00455, 1990.
Michelsen, N., van Geldern, R., Roßmann, Y., Bauer, I., Schulz, S., Barth, J. A. C., and Schüth, C.: Comparison of precipitation collectors used in isotope hydrology, Chemical Geology, 488, 171–179, https://doi.org/10.1016/j.chemgeo.2018.04.032, 2018.
Munksgaard, N. C., Wurster, C. M., Bass, A., and Bird, M. I.: Extreme short-term stable isotope variability revealed by continuous rainwater analysis, Hydrological Processes, 26, 3630–3634, https://doi.org/10.1002/hyp.9505, 2012.
Natali, S., Nigro, M., Baneschi, I., Giannecchini, R., Doveri, M., and Zanchetta, G.: On the reliability of tube-dip-in-water precipitation collectors in isotope hydrology: A field experiment for low rainfall amounts, Journal of Hydrology, 644, 132096, https://doi.org/10.1016/j.jhydrol.2024.132096, 2024.
Nigro, M., Žagar, K., and Vreča, P.: A Simple Water Sample Storage Test for Water Isotope Analysis, Sustainability, 16, 4740, https://doi.org/10.3390/su16114740, 2024.
Prechsl, U., Gilgen, A., Kahmen, A., and Buchmann, N.: Reliability and quality of water isotope data collected with a low-budget rain collector, Rapid communications in mass spectrometry: RCM, 28, 879–885, https://doi.org/10.1002/rcm.6852, 2014.
Radtke, C. F., Yang, X., Müller, C., Rouhiainen, J., Merz, R., Lutz, S. R., Benettin, P., Wei, H., and Knöller, K.: Nitrate and Water Isotopes as Tools to Resolve Nitrate Transit Times in a Mixed Land Use Catchment, Hydrol. Earth Syst. Sci. Discuss. [preprint], https://doi.org/10.5194/hess-2024-109, 2024.
Romañach, R., Castro, A., and Esbensen, K.: WHAT are sampling errors – and WHAT can we do about them? Part 1, Spectroscopy Europe, 36, https://doi.org/10.1255/sew.2021.a11, 2021.
Schürch, M., Kozel, R., Schotterer, U., and Tripet, J.-P.: Observation of isotopes in the water cycle – the Swiss National Network (NISOT), Environmental Geology, 45, 1–11, https://doi.org/10.1007/s00254-003-0843-9, 2003.
Terzer-Wassmuth, S., Araguás-Araguás, L. J., Copia, L., and Wassenaar, L. I.: High spatial resolution prediction of tritium (3H) in contemporary global precipitation, Scientific Reports, 12, 10271, https://doi.org/10.1038/s41598-022-14227-5, 2022.
Terzer-Wassmuth, S., Wassenaar, L. I., Welker, J. M., and Araguás-Araguás, L. J.: Improved high-resolution global and regionalized isoscapes of O, H and -excess in precipitation, Hydrological Processes, 35, e14254, https://doi.org/10.1002/hyp.14254, 2021.
von Freyberg, J., Knapp, J. L. A., Rücker, A., Studer, B., and Kirchner, J. W.: Technical note: Evaluation of a low-cost evaporation protection method for portable water samplers, Hydrol. Earth Syst. Sci., 24, 5821–5834, https://doi.org/10.5194/hess-24-5821-2020, 2020.
Weaver, J. and Talma, S.: Cumulative rainfall collectors – A tool for assessing groundwater recharge, Water SA, 31, https://doi.org/10.4314/wsa.v31i3.5216, 2007.
WMO: Evaporation Losses from Storage Gauges, edited by: Sevruk, B., in: Distribution of Precipitation in Mountainous Areas, Geilo Symposium, Norway, 31 July–5 August 1972, vol. II, Technical Papers, WMO-No. 326, World Meteorological Organization, Geneva, 96–102, 1972.





